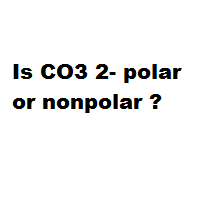
Section 11-1: Molecular Geometry: Using VSEPR Theory to Determine Three-Dimensional Shapes and Bond Angles

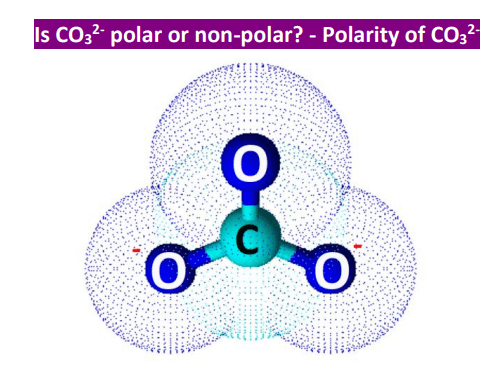
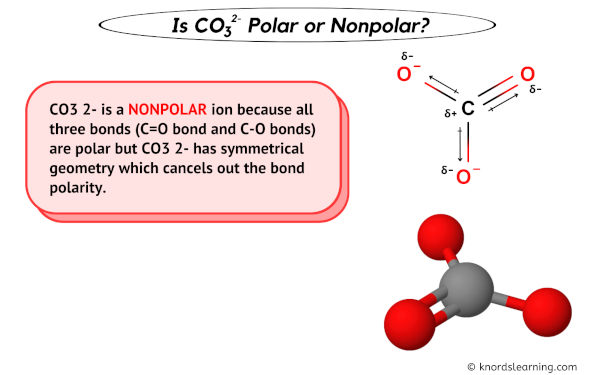
Geometry Of Molecules - Is CO3 2- Polar or Nonpolar (Carbonate ion) | Facebook | By Geometry Of Molecules | Is CO3 2- Polar or Nonpolar (Carbonate ion) Carbonate ion is a

Structure of dolomite MgCa(CO3)2 C-grey, Mg 2+ -blue, Ca 2+ -green, O... | Download Scientific Diagram
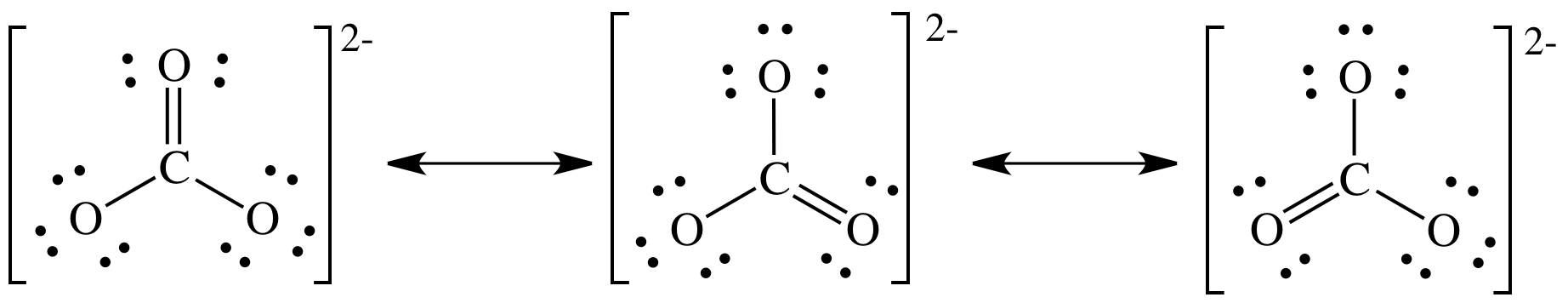
Can a molecule (like CO3 2-) be nonpolar even if it has two single bonds and one double bonds? - Quora
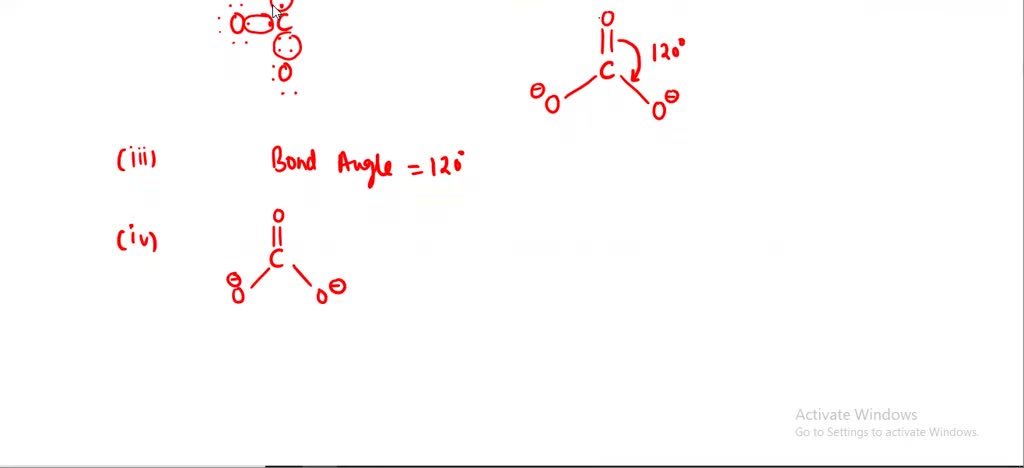


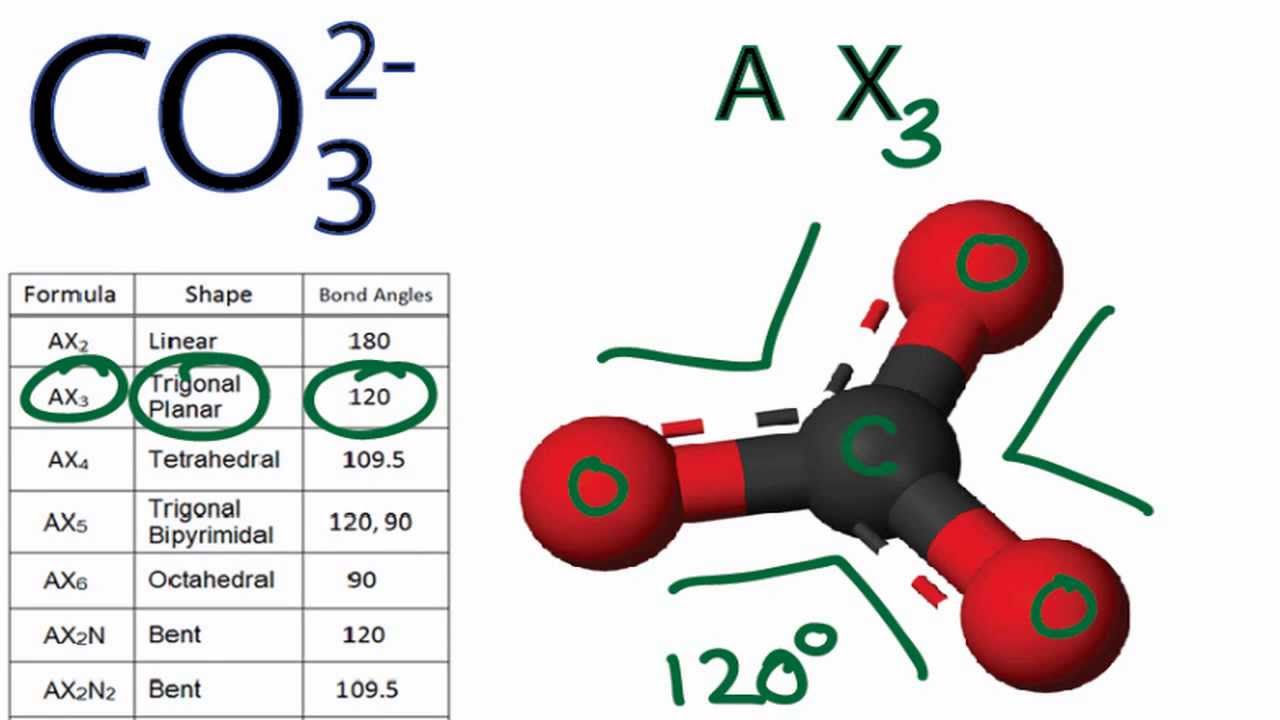



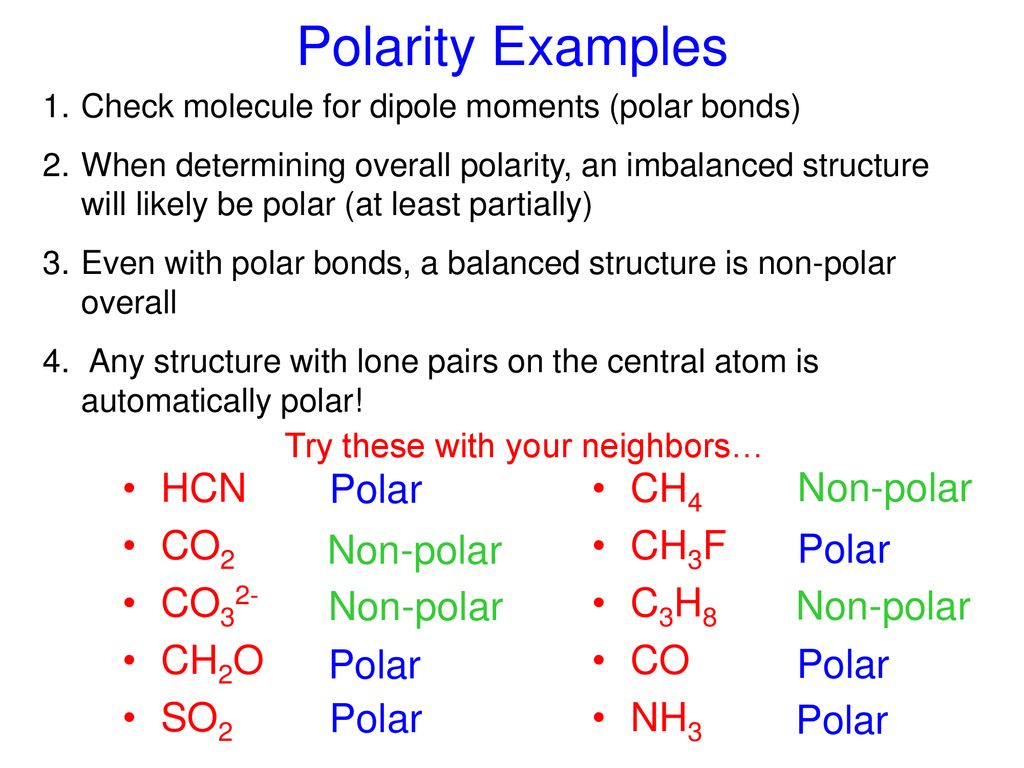


![SOLVED: Which of the following molecules is polar? [SO3]2 - [SbF6] - [PF4]+ [SiO4]4 - [CO3]2 - SOLVED: Which of the following molecules is polar? [SO3]2 - [SbF6] - [PF4]+ [SiO4]4 - [CO3]2 -](https://cdn.numerade.com/ask_images/726bca042e434fde93424aecaa925380.jpg)